Femara, Arimidex & Aromasin: The Aromatase Inhibitors
What are they?
There are three of these drugs currently in use in Australia in the treatment of breast cancer that is hormone sensitive (positive for the oestrogen receptor ("ER+") and/or progesterone receptor ("PR+"): letrozole ("Femara"), anastrozole ("Arimidex"), and exemestane ("Aromasin"). All are available on the Australian Pharmaceutical Benefits Scheme (PBS) for women after menopause for adjuvant therapy of early breast cancer and for management of metastatic ("secondary") breast cancer that is ER+ and/or PR+.
How do they work?
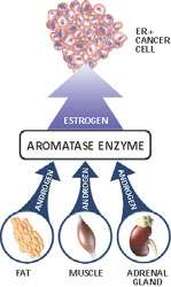
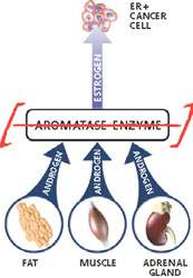
These drugs lower the levels of the active circulating female sex hormone, oestrogen, to almost zero. They are only used when the main source of oestrogen, the ovaries, have ceased producing the hormone, which happens naturally after menopause. After menopause, oestrogen is mainly produced in the fat tissue of the body and in tumour tissue itself by the action of an enzyme called “aromatase”. This enzyme acts on circulating steroid hormones called “androgens” which are produced in the adrenal glands, fat tissue and muscle (See Figure). Aromatase converts the androgens into active oestrogen. The aromatase inhibitors (AIs) block this enzyme virtually completely, thus depriving the body, and breast cancer cells, of active oestrogen. Letrozole and anastrozole are both synthetic non-steroid molecules, whereas exemestane is itself a steroid molecule.
AIs do not block the production of important adrenal hormones, like the androgens themselves and cortisone.
There are three of these drugs currently in use in Australia in the treatment of breast cancer that is hormone sensitive (positive for the oestrogen receptor ("ER+") and/or progesterone receptor ("PR+"): letrozole ("Femara"), anastrozole ("Arimidex"), and exemestane ("Aromasin"). All are available on the Australian Pharmaceutical Benefits Scheme (PBS) for women after menopause for adjuvant therapy of early breast cancer and for management of metastatic ("secondary") breast cancer that is ER+ and/or PR+.
How do they work?
These drugs lower the levels of the active circulating female sex hormone, oestrogen, to almost zero. They are only used when the main source of oestrogen, the ovaries, have ceased producing the hormone, which happens naturally after menopause. After menopause, oestrogen is mainly produced in the fat tissue of the body and in tumour tissue itself by the action of an enzyme called “aromatase”. This enzyme acts on circulating steroid hormones called “androgens” which are produced in the adrenal glands, fat tissue and muscle (See Figure). Aromatase converts the androgens into active oestrogen. The aromatase inhibitors (AIs) block this enzyme virtually completely, thus depriving the body, and breast cancer cells, of active oestrogen. Letrozole and anastrozole are both synthetic non-steroid molecules, whereas exemestane is itself a steroid molecule.
AIs do not block the production of important adrenal hormones, like the androgens themselves and cortisone.
When are they used?
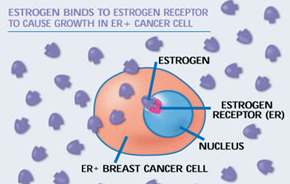
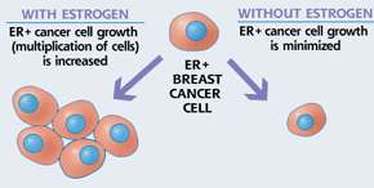
Aromatase inhibitors are used in post-menopausal women with advanced, or metastatic, breast cancer which is positive for the presence of the oestrogen receptor (ER)and/or progesterone receptor (See Figure). Presence of these receptors on the surface of breast cancer cells indicates that the cancer is "hormone sensitive". Over 70% of breast cancers are hormone sensitive.
They may also be indicated in adjuvant therapy of early breast cancer that is hormone sensitive (see below), but only in women who have been through the menopause. They do not work before menopause because the ovaries produce too much estrogen.
How are they taken?
Arimidex: 1 mg tablets taken once daily, before or after food.
Femara: 2.5 mg tablets taken once daily, before or after food.
Aromasin: 25 mg tablets taken once daily after food.
Is one better than the other?
The evidence for superiority of one over the other is slight. Claims to the contrary are mainly drug-company propaganda. Arimidex or Femara are currently considered the first choice in the treatment of ER +ve metastatic breast cancer. Clinical trials show them to be superior to tamoxifen in tumour response rates and time to tumour progression. Aromasin is only licensed in Australia for use in ER + metastatic breast cancer after the drug tamoxifen has failed to work.
Aromasin is the only one that will give responses after failure of one of the other aromatase inhibitors, because it works slightly differently to cause irreversible inhibition of the enzyme.
Margaret (not her real name or picture) |
What are their side-effects?
These drugs were generally well tolerated across all studies as first-line and second-line treatment for advanced breast cancer. Approximately one-third of the patients treated can be expected to experience mild adverse reactions. The most frequently reported adverse reactions in the clinical trials were hot flushes, nausea and hair thinning. Many adverse reactions can be attributed to the normal consequences of oestrogen deprivation (e.g. hot flushes, hair thinning).
My own experience is that side-effects are extremely uncommon, and I have certainly not seen them in one-third of my patients - more like one-in-ten.
Reported side-effects include:
They should not be taken by women who are still having their menstrual periods, during pregnancy or breast-feeding.
What if I forget to take it one day?
Don’t worry: just take it the next day as normal. The drug lasts in the blood-stream for around 2 days. But try to be regular with the daily dosing.
Adjuvant treatment of early breast cancer: Are these drugs alternatives to tamoxifen?
The aromatase inhibitors are being increasingly used in preference to tamoxifen in post-menopausal women.
The main trial in early breast cancer is the ATAC (Arimidex, Tamoxifen, Alone or in Combination) Trial. In this trial, over 9000 women were randomised in a double-blind fashion (neither the women nor the investigators knew who was getting what, to avoid any sort of bias). They received either tamoxifen, anastrazole or both drugs together. The study shows that, with over 8 years of median follow-up time, in the anastrazole-only arm, when compared to the other two arms of the trial, there were statistically significant data to support:
Use of aromatase inhibitors in pre-menopausal women - the SOFT and TEXT trials.
Recent data suggest that the use of aromatase inhibitors in women who are premenopausal is beneficial in comparison to Tamoxifen. AIs are only given to premenopausal women in conjunction with goserelin (Zoladex), which is a monthly injection given to suppress ovarian function. This monthly injection effectively makes a premenopausal woman, post-menopausal. You can read more about Zoladex here. Whilst the data from the SOFT and TEXT studies is robust, decisions about implementing this approach in mainstream early breast cancer treatment need to made with serious consideration. While a lot of young women tolerate Zoladex well, there are many who don't, and side effects such as hot flushes, changes to sexual drive and function as well as the vaginal environment can severely impact quality of life.
Jenny and I have attended many meetings where the implementation of such treatment is a topic of hot debate. Some medical oncologists argue that because the data is so compelling, Zoladex plus an AI should be routinely used in premenopausal women who have high risk disease, regardless of the side effects caused. Other medical oncologists argue that a young woman's quality of life is more important, particularly if the benefit of the combination is only small (less than 10%). My opinion is that this treatment should be considered in all high risk premenopusal women, but only after a discussion between your medical oncologist and yourself.
You can read the latest about the SOFT and TEXT studies here. The original published papers are listed below:
Adjuvant Ovarian Suppression in Premenopausal Breast Cancer
Adjuvant Exemestane with Ovarian Suppression in Premenopausal Breast Cancer
Absolute Benefit of Adjuvant Endocrine Therapies for Premenopausal Women With Hormone Receptor–Positive, Human Epidermal Growth Factor Receptor 2–Negative Early Breast Cancer: TEXT and SOFT Trials
These drugs were generally well tolerated across all studies as first-line and second-line treatment for advanced breast cancer. Approximately one-third of the patients treated can be expected to experience mild adverse reactions. The most frequently reported adverse reactions in the clinical trials were hot flushes, nausea and hair thinning. Many adverse reactions can be attributed to the normal consequences of oestrogen deprivation (e.g. hot flushes, hair thinning).
My own experience is that side-effects are extremely uncommon, and I have certainly not seen them in one-third of my patients - more like one-in-ten.
Reported side-effects include:
- Hot flashes ("hot flushes")
- Nausea, vomiting, constipation, loss of appetite.
- Hair thinning (For further information, click here)
- Osteoporosis. This is a potentially serious side-effect, especially if the agents are being used over several years as part of the adjuvant treatment of early breast cancer. There is more information on osteoporosis and its management here. The most serious consequence of osteoporosis is bone fractures, especially in the spine and hips. The overall risk of these happening in the ATAC Trial (see below) was 7.1%. Because of this risk, all women likely to be taking aromatase inhibitors for more than 12 months should have bone mineral density measurement as a baseline, and then at regular intervals, probably 2nd yearly, and should also follow all the preventative measures outlined here.
- Rashes
- Muscle and joint discomfort. This is quite common (around 20%) but variable in intensity. In the worst cases, women typically say something like "I feel like I'm suddenly very old and creaky. All my joints are stiff. My hands are stiff, especially in the morning. I find it hard to get my knees to get going when I stand up." This problem seems to be at its worst about the second month of treatment and then eases off. Sometimes a change from one aromatase inhibitor to another will cause less problems.
- Fluid retention
- Mood disturbance
- Sexual dysfunction, including loss of libido and vaginal dryness
- Thrombophlebitis (inflammation with clotting in the surface veins of the limbs) (less common than with tamoxifen)
- Fatigue
- Dizziness
They should not be taken by women who are still having their menstrual periods, during pregnancy or breast-feeding.
What if I forget to take it one day?
Don’t worry: just take it the next day as normal. The drug lasts in the blood-stream for around 2 days. But try to be regular with the daily dosing.
Adjuvant treatment of early breast cancer: Are these drugs alternatives to tamoxifen?
The aromatase inhibitors are being increasingly used in preference to tamoxifen in post-menopausal women.
The main trial in early breast cancer is the ATAC (Arimidex, Tamoxifen, Alone or in Combination) Trial. In this trial, over 9000 women were randomised in a double-blind fashion (neither the women nor the investigators knew who was getting what, to avoid any sort of bias). They received either tamoxifen, anastrazole or both drugs together. The study shows that, with over 8 years of median follow-up time, in the anastrazole-only arm, when compared to the other two arms of the trial, there were statistically significant data to support:
- Fewer recurrences of breast cancer
- Improved disease-free survival
- Fewer hot flashes (also called hot flushes)
- Less vaginal bleeding
- Less cancer of the lining of the womb (endometrial cancer)
- Less blood clotting (thromobotic) events
- More arthritis
- More bone fractures from osteoporosis during the period of five years that the drugs were administered, but not after they were ceased
- No difference in overall survival
Use of aromatase inhibitors in pre-menopausal women - the SOFT and TEXT trials.
Recent data suggest that the use of aromatase inhibitors in women who are premenopausal is beneficial in comparison to Tamoxifen. AIs are only given to premenopausal women in conjunction with goserelin (Zoladex), which is a monthly injection given to suppress ovarian function. This monthly injection effectively makes a premenopausal woman, post-menopausal. You can read more about Zoladex here. Whilst the data from the SOFT and TEXT studies is robust, decisions about implementing this approach in mainstream early breast cancer treatment need to made with serious consideration. While a lot of young women tolerate Zoladex well, there are many who don't, and side effects such as hot flushes, changes to sexual drive and function as well as the vaginal environment can severely impact quality of life.
Jenny and I have attended many meetings where the implementation of such treatment is a topic of hot debate. Some medical oncologists argue that because the data is so compelling, Zoladex plus an AI should be routinely used in premenopausal women who have high risk disease, regardless of the side effects caused. Other medical oncologists argue that a young woman's quality of life is more important, particularly if the benefit of the combination is only small (less than 10%). My opinion is that this treatment should be considered in all high risk premenopusal women, but only after a discussion between your medical oncologist and yourself.
You can read the latest about the SOFT and TEXT studies here. The original published papers are listed below:
Adjuvant Ovarian Suppression in Premenopausal Breast Cancer
Adjuvant Exemestane with Ovarian Suppression in Premenopausal Breast Cancer
Absolute Benefit of Adjuvant Endocrine Therapies for Premenopausal Women With Hormone Receptor–Positive, Human Epidermal Growth Factor Receptor 2–Negative Early Breast Cancer: TEXT and SOFT Trials