Anti-HER2 therapies
What is HER2?
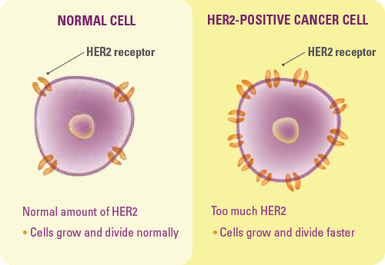
Human Epidermal Growth Factor Receptor 2 (HER2) is a protein which helps cells grow and divide. The HER2 gene produces HER2 proteins, and these are found as receptors on the surface of some breast cancer cells. Some people with breast cancer have higher than normal levels of HER2, and these are known as HER2 positive breast cancers. Approximately one in five breast cancers are HER2-positive.
Tumours are tested for HER2 as they are for the oestrogen and progesterone receptors. There are two methods commonly used for testing HER2, immunohistochemistry (IHC) and in situ hybridisation (ISH) testing. IHC testing is done routinely and determines how much of the HER2 protein is within the cancer cell. Results are either 1+ (negative), 2+ (equivocal) or 3+ (positive). ISH testing is done on request (we request it for all patients) and is a confirmatory test which determines if there is an excess of the HER2 gene within the cancer cell. Results are either positive (HER2 gene amplification) or negative (no HER2 gene amplification).
All of the below treatments are available for patients with either HER2 positive early breast cancer or HER2 positive metastatic breast cancer. They are therapies which specifically target the HER2 receptor on the surface of the cancer cell. For patients who don't over-express HER2, they are useless and they simply do not work.
Herceptin (Trastuzumab)
Herceptin works by targeting the HER2 receptor and blocking it's action. It is currently on the PBS for both early and metastatic breast cancer.
In the early setting, Herceptin is most commonly given 3 weekly for a year (17 doses). It is given via intravenous infusion over 90 minutes (initial dose) or 30 minutes (subsequent doses).
For patients with metastatic breast cancer (spread to distant parts of the body), Herceptin is given three weekly via intravenous infusion for as long as it keeps the cancer under control and is well tolerated. We have had some patients on Herceptin for more than 5 years!
In the USA, there is also a formulation of Herceptin which allows it to be given via subcutaneous injection. Conversations with the PBS here in Australia are ongoing, but I'm sure we will see this available in the not too distant future.
Side effects
Herceptin is generally very well tolerated. It does not cause nausea, hair loss, or low blood counts.
The following are important reported side-effects:
Fevers and rigors (chills)(Common)
This is usually associated with the first actual infusion, and may require treatment with paracetamol, anti-histamines or pethidine injection. Paracetamol is routinely given as pre-medication for the first four treatments.
Severe allergic reactions (Rare)
Severe hypersensitivity reactions including anaphylactic shock have occurred on rare occasions and have even caused death. These occur during the actual infusion. Any sign of difficulty breathing or facial or throat swelling requires cessation of infusion, and administration of antihistamines and corticosteroids, like dexamethasone.
Heart dysfunction (cardiomyopathy)(Rare)
Signs and symptoms of heart dysfunction, such as breathlessness, increased cough, and swollen calves have been observed in patients treated with Herceptin. Congestive heart failure associated with Herceptin therapy may be severe and has been associated with disabling cardiac failure, death, and heart wall thrombosis leading to stroke. These affects are minimised by careful cardiac monitoring and you will require regular scanning of the heart by echocardiogram. Cardiac dysfunction was more common when Herceptin was used in combination with anthracyclines like Adriamycin. We no longer use that combination. The majority of patients with heart dysfunction from Herceptin improve on its withdrawal.
More information may be found on the eviQ patient information sheet.
Tykerb (Lapatinib)
Lapatinib (Tykerb®) is a drug, like trastuzumab, which blocks the activity of the HER2 protein, but it does so by a different mechanism. Lapatinib binds to the part of the protein found inside breast cancer cells. It also blocks a related growth factor receptor called EGFR.
It is currently on the PBS for patients with metastatic breast cancer in combination with the oral chemotherapy agent, Capecitabine (Xeloda). It is taken every day, the recommended dosage is 1250mg daily (5 tablets).
It is only available in combination with Xeloda because the original clinical trial which found Lapatinib to be an effective treatment showed that HER2 positive patients who received treatment with Lapatinib plus Xeloda had a longer time before their disease progressed in comparison to those who had Xeloda alone.
For more information on the combination of Tykerb and Xeloda, click here.
Perjeta (Pertuzumab)
Pertuzumab is a humanized monoclonal antibody being studied in early and advanced stages of HER2-positive breast cancer and advanced HER2-positive gastric cancer. Pertuzumab is unique in that it is designed specifically to prevent the HER2 receptor from pairing with other HER receptors (EGFR/HER1, HER3 and HER4), a process that is believed to play an important role in the growth and formation of several different cancer types. By preventing receptor pairing, pertuzumab is thought to block cell signalling, which may inhibit cancer cell growth or lead to the death of the cancer cell. Binding of pertuzumab to HER2 may also signal the body’s immune system to destroy the cancer cells.
In February 2015 results were published from CLEOPATRA, (CLinical Evaluation Of Pertuzumab And TRAstuzumab) the first randomised Phase III study of the investigational HER2-targeted medicine pertuzumab in the New England Journal of Medicine. The study compared the combination of pertuzumab, Herceptin (trastuzumab) and docetaxel chemotherapy to Herceptin and docetaxel alone in people with previously untreated HER2-positive metastatic breast cancer (mBC). People who received pertuzumab in combination with Herceptin and chemotherapy experienced a 38 percent reduction in the risk of their disease worsening or death (progression-free survival, or PFS), (Hazards Ratio=0.62; p-value=<0.0001). The median PFS improved by 6.3 months from 12.4 months for Herceptin and chemotherapy to 18.7 months for pertuzumab, Herceptin and chemotherapy. The median overall survival was 56.5 months in the pertuzumab group and 40.8 months in the control group. This means that the patients in the pertuzumab group survived 15.7 months longer (on average) than those who did not receive pertuzumab. This was a statistically significant finding.
The mechanisms of action of pertuzumab and Herceptin are believed to complement each other as both bind to the HER2 receptor but on different regions. This is believed to provide a more comprehensive blockade of HER signalling pathways.
On the basis of this study, pertuzumab is now available on the PBS for patients with metastatic breast cancer. It is only available in the first-line setting i.e. patients who have not yet had any treatment for their HER2 positive MBC.
It is given via intravenous infusion once every three weeks over 60 minutes (initial infusion) or 30 minutes (second and subsequent infusions) in combination with Herceptin and docetaxel (Taxotere).
More information about Taxotere, Perjeta and Herceptin can be found here.
Kadcyla (Trastuzumab emtansine or TDM1)
Trastuzumab emtansine is a clever agent which is made up of two anti-cancer drugs combined into one. It is a monoclonal antibody combined with a microtubule inhibitory agent DM1. The DM1 (chemo-like drug) is delivered to the cancer cell via the monoclonal antibody component which binds to the HER2 receptor on the surface of the cancer cell. Once the antibody is bound to the cancer cell, the DM1 component kills it. A video on how it works can be found here.
In January 2015, the results from the EMILIA study were published in the New England Journal of Medicine. It looked at the differences between two groups of HER2 positive patients with metastatic breast cancer who were randomly assigned to receive either TDM1 or Lapatinib and Capecitabine (Tykerb / Xeloda - see above). All patients had been previously treated with Trastuzumab (Herceptin) and a taxane (type of chemotherapy). The study found that treatment with TDM1 significantly prolonged progression free and overall survival in comparison to Tykerb & Xeloda. Patients who received treatment with TDM1 progressed after 9.6 months versus 6.4 months in the patients who had Tykerb & Xeloda, and patients who had TDM1 lived 5.8 months (on average) longer than those who didn't. Both of these findings were statistically significant and, like Pertuzumab, it is now available on the PBS for women with metastatic breast cancer. The PBS has certain rules about which metastatic breast cancer patients can get TDM1 and which cannot. It is rather complicated, but if you fit the criteria we will offer this to you when or if the time arises.
TDM1 is given via intravenous infusion over 90 minutes (initial infusion) or 30 minutes (subsequent infusions) once every three weeks. Information regarding the side effects of TDM1 can be found on the eviQ patient information sheet.
Human Epidermal Growth Factor Receptor 2 (HER2) is a protein which helps cells grow and divide. The HER2 gene produces HER2 proteins, and these are found as receptors on the surface of some breast cancer cells. Some people with breast cancer have higher than normal levels of HER2, and these are known as HER2 positive breast cancers. Approximately one in five breast cancers are HER2-positive.
Tumours are tested for HER2 as they are for the oestrogen and progesterone receptors. There are two methods commonly used for testing HER2, immunohistochemistry (IHC) and in situ hybridisation (ISH) testing. IHC testing is done routinely and determines how much of the HER2 protein is within the cancer cell. Results are either 1+ (negative), 2+ (equivocal) or 3+ (positive). ISH testing is done on request (we request it for all patients) and is a confirmatory test which determines if there is an excess of the HER2 gene within the cancer cell. Results are either positive (HER2 gene amplification) or negative (no HER2 gene amplification).
All of the below treatments are available for patients with either HER2 positive early breast cancer or HER2 positive metastatic breast cancer. They are therapies which specifically target the HER2 receptor on the surface of the cancer cell. For patients who don't over-express HER2, they are useless and they simply do not work.
Herceptin (Trastuzumab)
Herceptin works by targeting the HER2 receptor and blocking it's action. It is currently on the PBS for both early and metastatic breast cancer.
In the early setting, Herceptin is most commonly given 3 weekly for a year (17 doses). It is given via intravenous infusion over 90 minutes (initial dose) or 30 minutes (subsequent doses).
For patients with metastatic breast cancer (spread to distant parts of the body), Herceptin is given three weekly via intravenous infusion for as long as it keeps the cancer under control and is well tolerated. We have had some patients on Herceptin for more than 5 years!
In the USA, there is also a formulation of Herceptin which allows it to be given via subcutaneous injection. Conversations with the PBS here in Australia are ongoing, but I'm sure we will see this available in the not too distant future.
Side effects
Herceptin is generally very well tolerated. It does not cause nausea, hair loss, or low blood counts.
The following are important reported side-effects:
Fevers and rigors (chills)(Common)
This is usually associated with the first actual infusion, and may require treatment with paracetamol, anti-histamines or pethidine injection. Paracetamol is routinely given as pre-medication for the first four treatments.
Severe allergic reactions (Rare)
Severe hypersensitivity reactions including anaphylactic shock have occurred on rare occasions and have even caused death. These occur during the actual infusion. Any sign of difficulty breathing or facial or throat swelling requires cessation of infusion, and administration of antihistamines and corticosteroids, like dexamethasone.
Heart dysfunction (cardiomyopathy)(Rare)
Signs and symptoms of heart dysfunction, such as breathlessness, increased cough, and swollen calves have been observed in patients treated with Herceptin. Congestive heart failure associated with Herceptin therapy may be severe and has been associated with disabling cardiac failure, death, and heart wall thrombosis leading to stroke. These affects are minimised by careful cardiac monitoring and you will require regular scanning of the heart by echocardiogram. Cardiac dysfunction was more common when Herceptin was used in combination with anthracyclines like Adriamycin. We no longer use that combination. The majority of patients with heart dysfunction from Herceptin improve on its withdrawal.
More information may be found on the eviQ patient information sheet.
Tykerb (Lapatinib)
Lapatinib (Tykerb®) is a drug, like trastuzumab, which blocks the activity of the HER2 protein, but it does so by a different mechanism. Lapatinib binds to the part of the protein found inside breast cancer cells. It also blocks a related growth factor receptor called EGFR.
It is currently on the PBS for patients with metastatic breast cancer in combination with the oral chemotherapy agent, Capecitabine (Xeloda). It is taken every day, the recommended dosage is 1250mg daily (5 tablets).
It is only available in combination with Xeloda because the original clinical trial which found Lapatinib to be an effective treatment showed that HER2 positive patients who received treatment with Lapatinib plus Xeloda had a longer time before their disease progressed in comparison to those who had Xeloda alone.
For more information on the combination of Tykerb and Xeloda, click here.
Perjeta (Pertuzumab)
Pertuzumab is a humanized monoclonal antibody being studied in early and advanced stages of HER2-positive breast cancer and advanced HER2-positive gastric cancer. Pertuzumab is unique in that it is designed specifically to prevent the HER2 receptor from pairing with other HER receptors (EGFR/HER1, HER3 and HER4), a process that is believed to play an important role in the growth and formation of several different cancer types. By preventing receptor pairing, pertuzumab is thought to block cell signalling, which may inhibit cancer cell growth or lead to the death of the cancer cell. Binding of pertuzumab to HER2 may also signal the body’s immune system to destroy the cancer cells.
In February 2015 results were published from CLEOPATRA, (CLinical Evaluation Of Pertuzumab And TRAstuzumab) the first randomised Phase III study of the investigational HER2-targeted medicine pertuzumab in the New England Journal of Medicine. The study compared the combination of pertuzumab, Herceptin (trastuzumab) and docetaxel chemotherapy to Herceptin and docetaxel alone in people with previously untreated HER2-positive metastatic breast cancer (mBC). People who received pertuzumab in combination with Herceptin and chemotherapy experienced a 38 percent reduction in the risk of their disease worsening or death (progression-free survival, or PFS), (Hazards Ratio=0.62; p-value=<0.0001). The median PFS improved by 6.3 months from 12.4 months for Herceptin and chemotherapy to 18.7 months for pertuzumab, Herceptin and chemotherapy. The median overall survival was 56.5 months in the pertuzumab group and 40.8 months in the control group. This means that the patients in the pertuzumab group survived 15.7 months longer (on average) than those who did not receive pertuzumab. This was a statistically significant finding.
The mechanisms of action of pertuzumab and Herceptin are believed to complement each other as both bind to the HER2 receptor but on different regions. This is believed to provide a more comprehensive blockade of HER signalling pathways.
On the basis of this study, pertuzumab is now available on the PBS for patients with metastatic breast cancer. It is only available in the first-line setting i.e. patients who have not yet had any treatment for their HER2 positive MBC.
It is given via intravenous infusion once every three weeks over 60 minutes (initial infusion) or 30 minutes (second and subsequent infusions) in combination with Herceptin and docetaxel (Taxotere).
More information about Taxotere, Perjeta and Herceptin can be found here.
Kadcyla (Trastuzumab emtansine or TDM1)
Trastuzumab emtansine is a clever agent which is made up of two anti-cancer drugs combined into one. It is a monoclonal antibody combined with a microtubule inhibitory agent DM1. The DM1 (chemo-like drug) is delivered to the cancer cell via the monoclonal antibody component which binds to the HER2 receptor on the surface of the cancer cell. Once the antibody is bound to the cancer cell, the DM1 component kills it. A video on how it works can be found here.
In January 2015, the results from the EMILIA study were published in the New England Journal of Medicine. It looked at the differences between two groups of HER2 positive patients with metastatic breast cancer who were randomly assigned to receive either TDM1 or Lapatinib and Capecitabine (Tykerb / Xeloda - see above). All patients had been previously treated with Trastuzumab (Herceptin) and a taxane (type of chemotherapy). The study found that treatment with TDM1 significantly prolonged progression free and overall survival in comparison to Tykerb & Xeloda. Patients who received treatment with TDM1 progressed after 9.6 months versus 6.4 months in the patients who had Tykerb & Xeloda, and patients who had TDM1 lived 5.8 months (on average) longer than those who didn't. Both of these findings were statistically significant and, like Pertuzumab, it is now available on the PBS for women with metastatic breast cancer. The PBS has certain rules about which metastatic breast cancer patients can get TDM1 and which cannot. It is rather complicated, but if you fit the criteria we will offer this to you when or if the time arises.
TDM1 is given via intravenous infusion over 90 minutes (initial infusion) or 30 minutes (subsequent infusions) once every three weeks. Information regarding the side effects of TDM1 can be found on the eviQ patient information sheet.